An Introduction to the Electronic Structure of Atoms and Molecules
Professor of Chemistry / McMaster University / Hamilton, Ontario
An Introduction to the Electronic Structure of Atoms and MoleculesProfessor of Chemistry / McMaster University / Hamilton, Ontario
|
The helium atom is a good example of a many-electron atom (that is, an atom which contains more than one electron). No fundamentally new problems are encountered whether we consider two or ten electrons, but a very important problem arises in passing from the one-electron to the two-electron case. To see what this problem is, consider all the potential interactions found in a helium atom. Again, consider the electrons to be point charges and "freeze" them at some instantaneous positions in space (Fig.4-1).
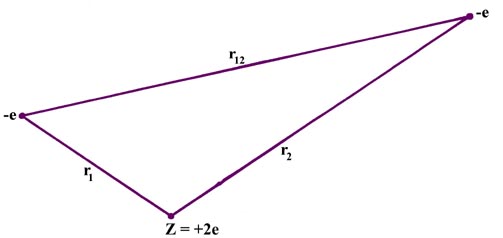
The potential energy, the average value of which is to be determined
by quantum mechanics, is
| (1) |
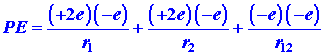 |
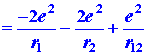 |
The first and second terms in equation (1) represent the attraction of the helium nucleus (for which Z = 2) for electrons 1 and 2 respectively. The last term represents the repulsion between the two electrons. It is this last term which makes the problem of the helium atom, and of all many-electron atoms, difficult to solve. No direct solution to the problem exists, the reason being that there are too many interactions to consider simultaneously. We must make some approximation in our approach to this problem.
 |
 |
 |
 |