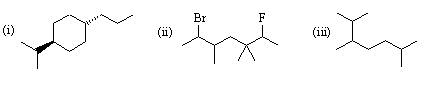
| Problem Set #4 | October 10, 1997 |
1. Give IUPAC names for the following structures:
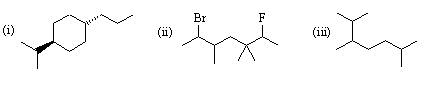
(a) 1-chloro-2,2-dimethylpentane
(b) 3-(1,1-dimethylethyl)hexane
(c) 4-propylheptane
(d) 2-bromo-3-chloro-5-(2-methylpropyl)dodecane
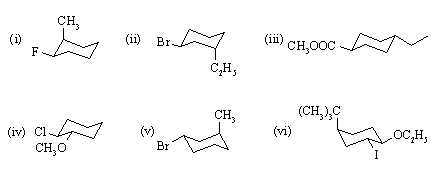
If a gauche OH-OH interaction is worth 1.6 kJ/mol, a gauche CH3-CH3 interaction 3.5 kJ/mol, and a gauche CH3-OH interaction 2.0 kJ/mol, how much more stable is your selected conformation above than the least stable conformer?
Invert the chair into the alternate chair. What is the orientation of the two C-Br bonds now? Identify any 1,3-diaxial interactions present in both conformers.
| Go to: |
Instructions for Printing this Document Answers Chem2O6 Problem Sets & Answers Chem2O6 Home Page. |
10oct97; wjl