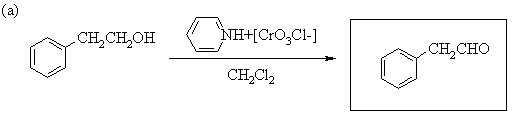
Chem2O6
- 1997/98| Assignment #3 | ANSWERS | January 12, 1998 |
1. Predict the principal products of the following reactions, showing stereochemistry where appropriate.
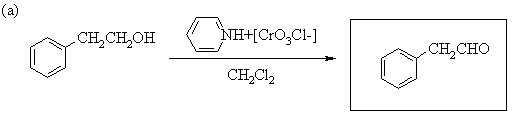
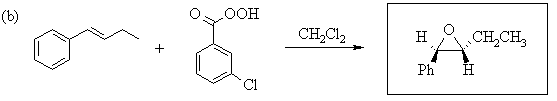
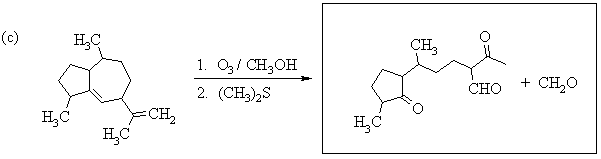
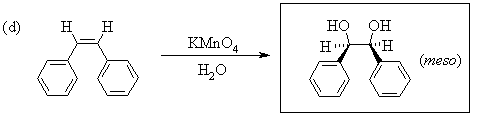
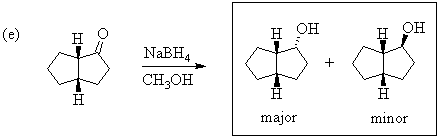
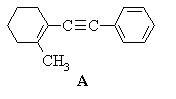
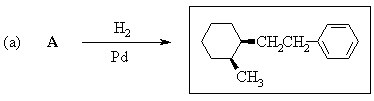
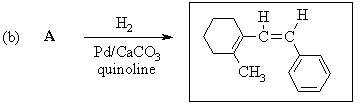
2. Predict the principal products of reaction of 1-methyl-2-(phenylethynyl)cyclohexene (A) with the following reagents, showing stereochemistry where appropriate:
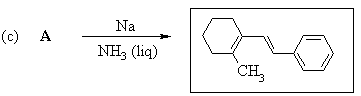
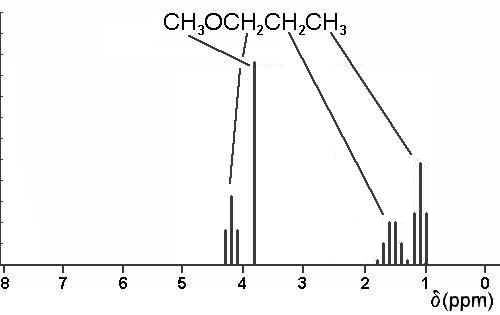
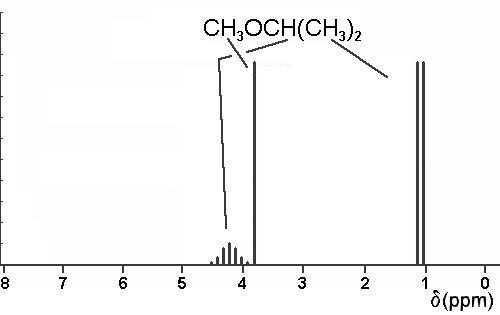
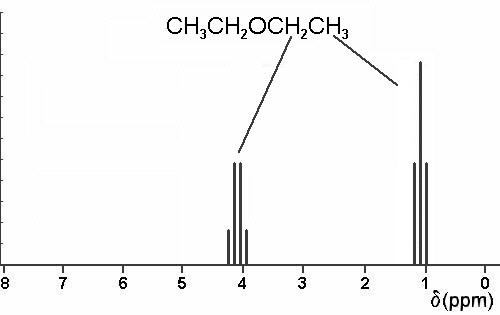
3. Using a crude graph, draw simple line sketches of the 1H nmr spectra expected for all the isomeric ethers of constitutional formula C4H10O. Expected chemical shifts should be accurate to within 0.5 ppm, and within each spectrum, the relative heights of the lines are to be proportional to the number of protons responsible for each of the predicted absorptions. Do not skew multiplets.
Just three structures are possible:
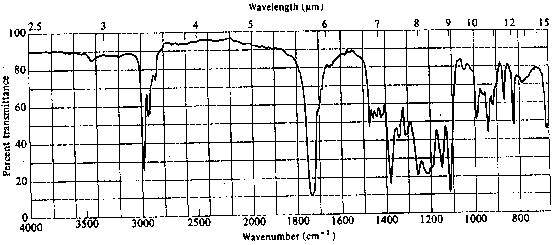
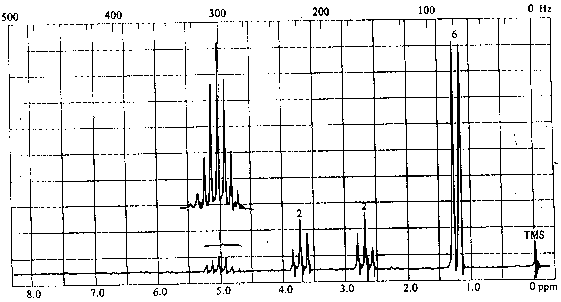
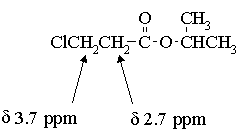
4. The infrared and 1H nmr spectra of a compound with molecular formula C6H11O2Cl are shown below. Deduce the structure of the compound from these data.
Molecular Formula
The molecular formula indicates there to be ONE site of unsaturation in the molecule.
Infrared Spectrum
The presence of a strong peak at ~1730 cm-1 clearly indicates that the molecule contains a carbonyl group. There are no OH, NH or vinylic C-H absorptions present. Aldehydes always show a pair of peaks at ~2700 and ~2900 cm-1; while the 2900 cm-1 peak is often difficult to pick out, there is clearly nothing at ~2700. Thus, the molecule is an aliphatic ketone, ester, or acid chloride. Esters typically show a strong absorption at ~1200 cm-1 due to the C-O stretch. There is absorption here, but it overlaps with a number of other bands and is difficult to assign reliably.
1H NMR Spectrum
The presence of the 1H septet at ~ 5ppm indicates that the two peaks at ~1.2 ppm are a doublet rather than two singlets; thus, the molecule contains an isopropyl group and a total of 4 types of hydrogens in relative intensities of 1:2:2:6.
The chemical shift of the isopropyl CH proton and the fact that the signal is not split further suggest that we have an isopropyl ester:
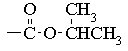
The two 2H triplets at 2.7 & 3.7 ppm indicates that the remaining C2H4Cl must be arranged as -CH2CH2Cl. Thus, the structure of the molecule is:
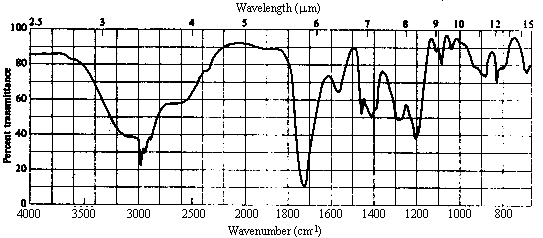
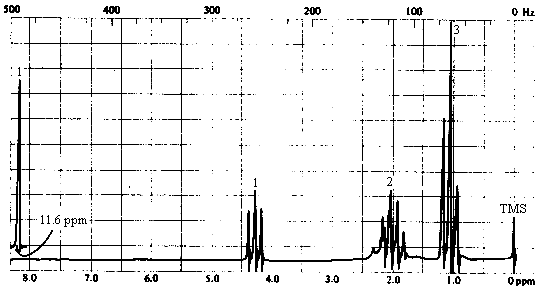
5. The infrared and 1H nmr spectra of a compound with molecular formula C4H7O2Cl are shown below. Note that the multiplet at d~2 ppm in the nmr spectrum is a pentet. Deduce the structure of the compound from these data.
| 1. Molecular Formula | C4H7ClO2 -> 1 site of unsaturation. |
| 2. Infrared Spectrum | - strong peak at ~1720 cm-1 -> C=O (this accounts for the site of unsaturation!) - strong, very broad absorption extending from 2500-3500 cm-1 -> OH. The broad range is characteristic of carboxylic acids (compare to the ir spectrum of 2-propanol in #3). |
| 1H NMR spectrum | - 4 types of H's: - singlet at 11.6 ppm (1H) -> aldehyde or carboxylic acid proton. The ir spectrum allows identification as a carboxylic acid. - triplet at ~4.2 ppm (1H) -> a single proton with 2 nearest neighbours. With only 3 carbons left, this must be due to a CH attached to a CH2! - triplet at 1 ppm (3H) -> three equivalent protons with 2 nearest neighbours. This must be due to a methyl group attached to a CH2. The last two bits of information, coupled with the fact that we have only C3H6Cl left, are only consistent with one structure - CHClCH2CH3!
- pentet at ~2 ppm (2H) -> two equivalent protons with 4 nearest neighbours. This is the
middle CH2 in the -CHClCH2CH3 chain. |
The only structure possible is:
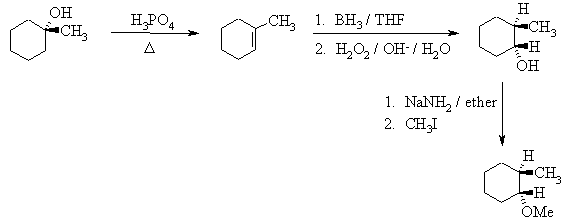
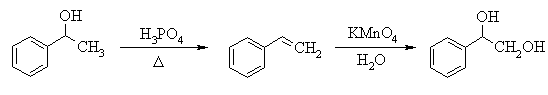
6. The following incomplete reaction sequences give only a starting material and a
product that can be prepared from it by a multistep reaction sequence. Show the reagents
that would that would be necessary to carry out each transformation, and any major
products that would be formed in the intermediate stages. 12jan98; wjl 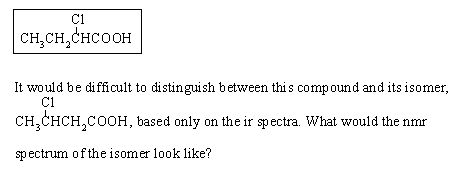
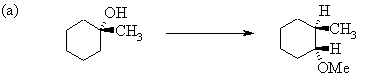
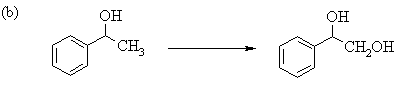

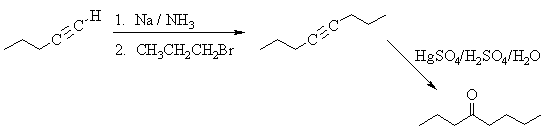
Go to:
Instructions for Printing this Document
Chem2O6 Problem Sets & Answers
Chem2O6 Home Page.