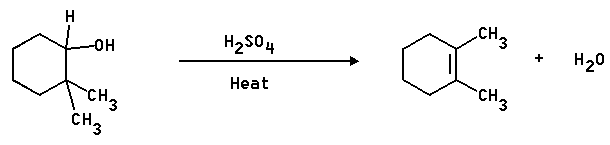
| Problem Set #10 | Answers | January 16, 1998 |
1. Propose a mechanism for the following reaction -
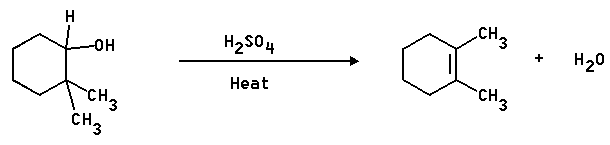
Answer:
2. Show how you would convert (R)-2-butanol to (S)-2-butanethiol?
Answer:
The conversion involves a change in configuration at C-2 of the butanol. In other words a bimolecular nucleophilic displacement (SN2) is needed.
SH- would be an effective nucleophile. This can be prepared by bubbling H2S into sodium hydroxide solution in water/ethanol.
BUT one cannot do a direct displacement of an OH group of an alcohol. This means that we must make the alcohol into a better leaving group. To do this we could react the 2-butanol with p-toluenesulphonyl chloride in pyridine as a solvent, isolate the tosylate ester, and then treat with SH- in water ethanol..........

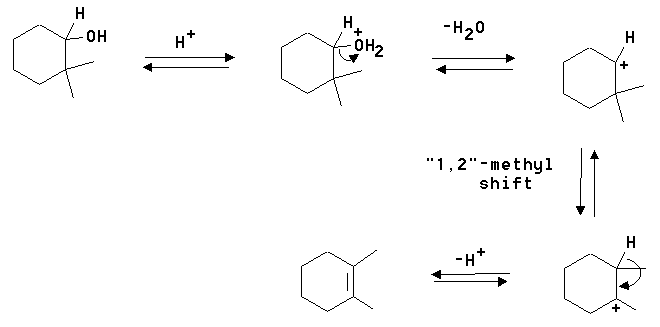
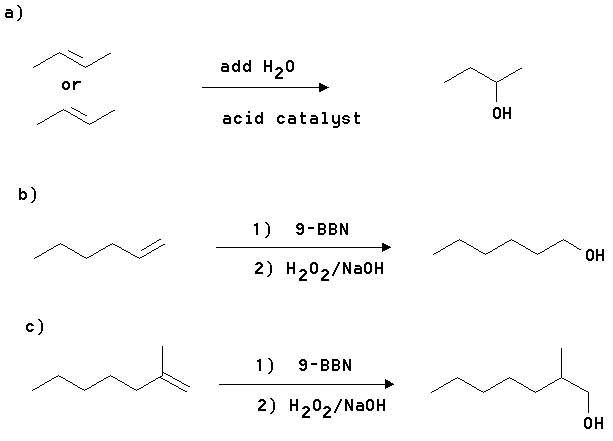
3. Give the structural formula of an alkene and show the reagents needed to make the following alcohols.
A) 2-Butanol
B) 1-Hexanol
C) 2-Methyl-1-heptanol
Answer:
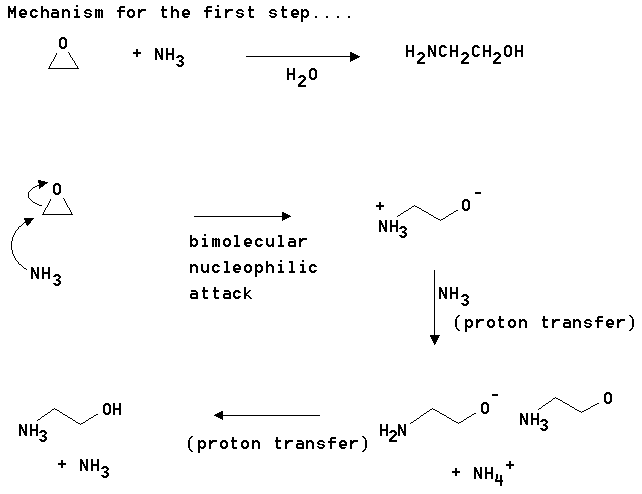
4. Triethanolamine (TEA) is used in industry to remove acid components from gas streams. It also finds extensive use as a buffer in biological systems. "TEA" is made from ammonia and ethylene oxide as shown below. Show how this synthesis occurs and propose a mechanism for one of the steps.
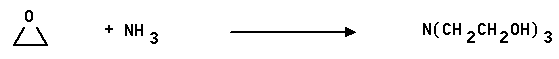
Answer:
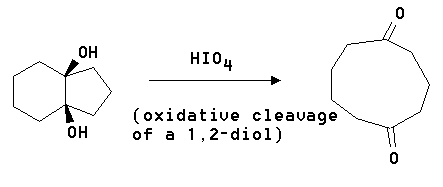
5. Show you would effect the following reaction.
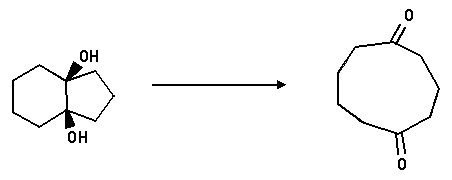
Answer:
6. Try question 12.43 in Ege.
| Go to: |
Instructions for Printing this Document Chem2O6 Problem Sets & Answers Chem2O6 Home Page. |
23jan98; jp