| Problem Set #12 | February 6, 1998 |
Ege has reams of exciting problems at the end of chapters 14 and 15. Try tackling, for example, the following.
14.41, 14. 39
15.21, 15.22, 15.25, 15.32, 15.34, 15.43, 15.44.
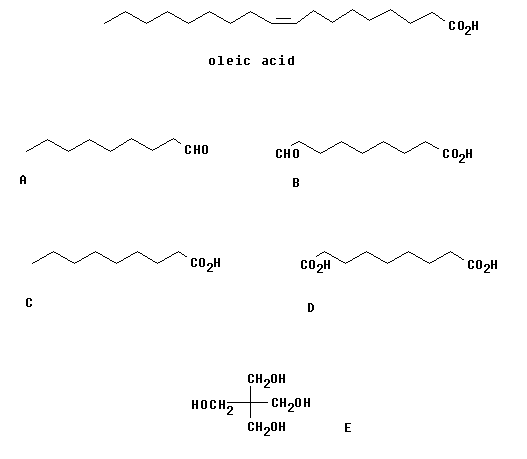
Oleic acid can be derived from a natural oils. In what form does it occur and what reaction is required to liberate oleic acid?
Oleic acid can be reacted to form the two products A and B shown below. What reagent would you use for this? What are the names of the two products?
The two products A and B are separated and then converted to the acids C and D, respectively. How would you carry out these transformations. Name C and D.
Diacid D can be reacted with 1-tridecanol to form the diester. Similarly, C can be reacted with E to form a tetra ester. How would you carry out these transformations?
These esters are used as components in synthetic lubricants. They are mixed with other components to form a lubricant which is said to give better engine wear. They can also be recycled easily.

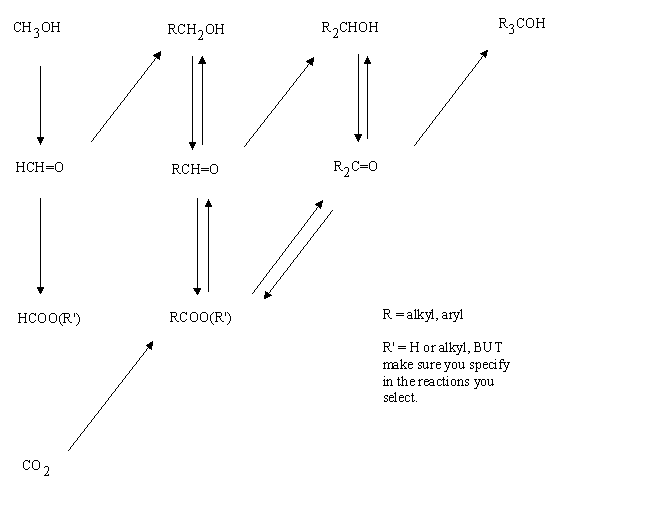
Transformations between the oxygen containing functional groups
Fill in the reagents and conditions needed to effect the various transformations
| Go to: |
Instructions for Printing this Document Chem2O6 Problem Sets & Answers Chem2O6 Home Page. |
6 feb 97; jp