![[Propellane Gradient Vector Field Map]](./f7b-201.gif)

![[Propellane Gradient Vector Field Map]](./f7b-201.gif) |
The second gradient vector field map in Figure 4 includes the trajectories, shown in bold, that both originate and terminate at the critical points found between nuclei that appear linked by a saddle in ![]() (r) in Figure 1. A critical point denotes an extremum in
(r) in Figure 1. A critical point denotes an extremum in ![]() (r), a point where
(r), a point where ![]()
![]() (r) = 0.
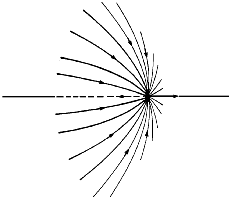
Associated with each such critical point is a set of trajectories that start at infinity and terminate at the critical point, only two of which appear in the symmetry plane shown in the figure. They define an interatomic surface, a surface that separates the basins of neighbouring atoms. There is a unique pair of trajectories that originate at each such critical point and terminate, one each, at the neighbouring nuclei. They define a line through space along which the electron density is a maximum. The two sets of trajectories associated with such a critical point, a bond critical point, the set that terminates at the critical point and defines the interatomic surface and the pair that originates there and defines the line of maximum density, are shown in Figure 5.
(r) = 0.
Associated with each such critical point is a set of trajectories that start at infinity and terminate at the critical point, only two of which appear in the symmetry plane shown in the figure. They define an interatomic surface, a surface that separates the basins of neighbouring atoms. There is a unique pair of trajectories that originate at each such critical point and terminate, one each, at the neighbouring nuclei. They define a line through space along which the electron density is a maximum. The two sets of trajectories associated with such a critical point, a bond critical point, the set that terminates at the critical point and defines the interatomic surface and the pair that originates there and defines the line of maximum density, are shown in Figure 5.
Figure 5. A three-dimensional display of the set of trajectories of |
|
In an equilibrium geometry the line of maximum density is called a bond path because the set of bond paths for a given molecule, the molecular graph, faithfully recovers the network of chemical bonds that are assigned on the basis of chemical considerations. Thus a pair of bonded atoms are linked by a line along which the electron density, the glue of chemistry, is maximally concentrated. Molecular structures predicted by the molecular graphs determined by the electron density are shown in Figure 6.
Figure 6. Molecular graphs - lines of maximum electron density linking bonded nuclei - in hydrocarbon molecules in diagrams 1 through 26, and boranes and carboranes below these. Bond critical points, where the trajectories defining the bond path originate, are denoted by dots. Note that the bond paths can be curved away from the internuclear axis in strained or in electron deficient molecules. A molecular graph and the characteristics of the density at the bond critical points provide a concise summary of the bonding within a molecule or crystal.
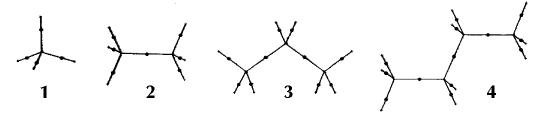
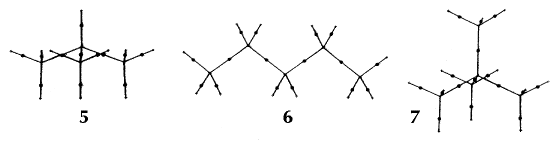
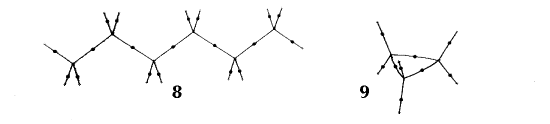
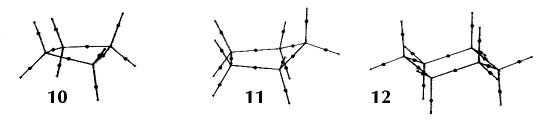
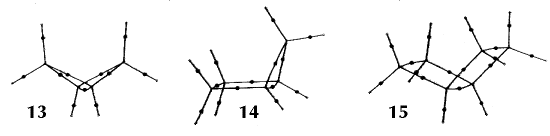
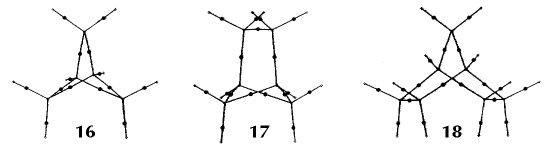
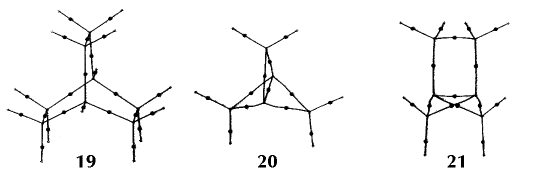
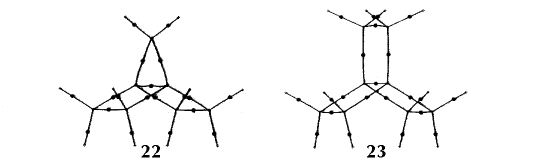
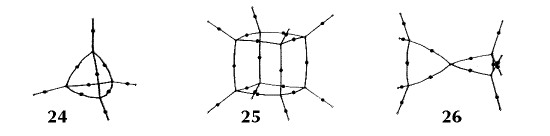
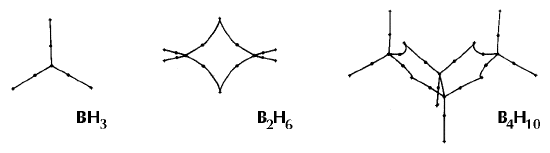
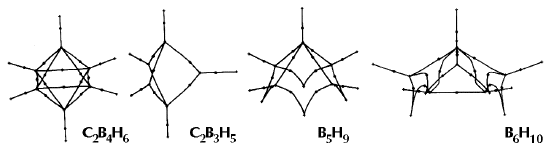
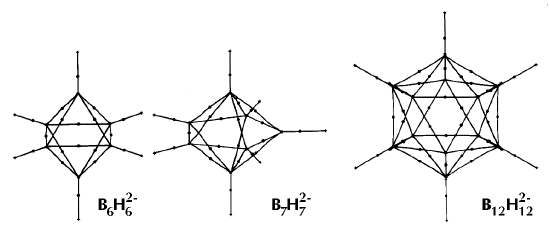
The molecular graph undergoes discontinuous and abrupt changes if the nuclei are displaced into critical configurations. When this occurs, one makes or breaks certain of the bonds and changes one structure into another. These changes are described and predicted using the mathematics of qualitative dynamics and the resulting theory of structural stability is illustrated in Figure 7 for the very strained molecule called [1,1,1]propellane.
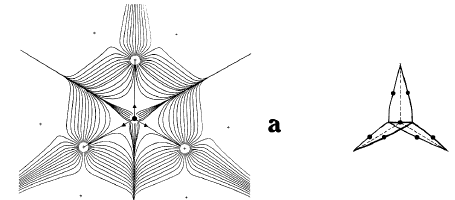
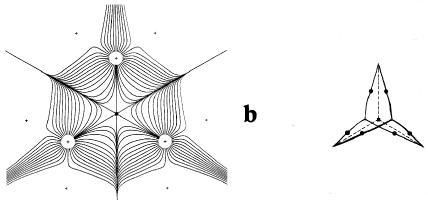
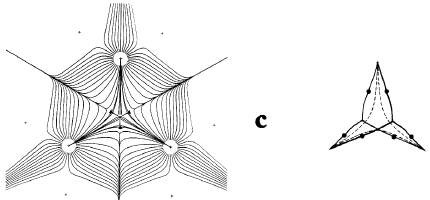
Figure 7. Diagrams illustrating changes in structure induced by the dynamics of the nuclei. The molecular graph in a is for the highly strained [1,1,1]propellane molecule, C5H6 (the two hydrogens attached to each apical carbon atom are not indicated). The gradient vector field maps are for the symmetry plane containing the C-C bridgehead bond critical point and the three apical carbon atoms. When the separation between the two bridgehead nuclei is increased to a critical value, the bond critical point coalesces with the three neighbouring ring critical points to form a singularity in(r), as depicted by the gradient vector field map in b. The singularity is unstable and its formation signifies the breaking of the C-C bridgehead bond. Further separation of the nuclei causes it to bifurcate into a cage critical point yielding a new structure in which the bridgehead carbon atoms are not bonded to one another, the cage structure depicted in c.


Next Section: The Topological Atom is the Quantum Atom
Previous Section: What is an Atom?
| Go to: |
R.F.W. Bader Research Group AIMPAC and Other AIM Software Department of Chemistry Faculty Department of Chemistry Welcome Page McMaster University Welcome Page |
02dec97; rfwb