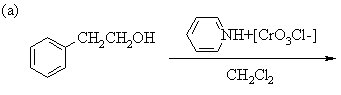
Chem2O6
- 1997/98| Assignment #3 | Due: January 9, 1998 |
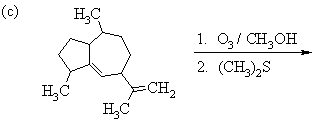
1. Predict the principal products of the following reactions, showing stereochemistry where appropriate.
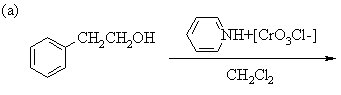
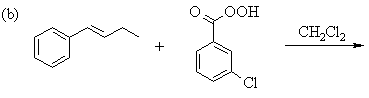
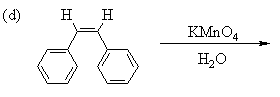
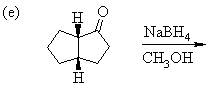
2. Predict the principal products of reaction of 1-methyl-2-(phenylethynyl)cyclohexene (A) with the following reagents, showing stereochemistry where appropriate:
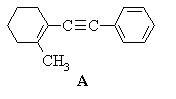
(a) H2 / Pd
(b) H2 / Pd / CaCO3 / quinoline
(c) Li / NH3(liq)
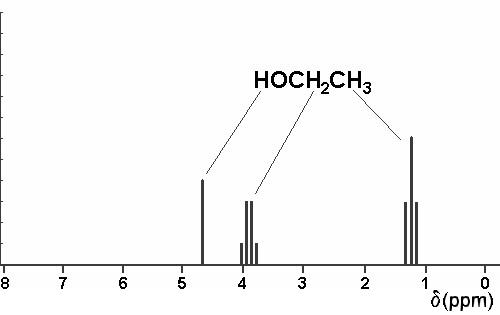
3. Using a crude graph, draw simple line sketches of the 1H nmr spectra expected for all the isomeric ethers of constitutional formula C4H10O. Expected chemical shifts should be accurate to within 0.5 ppm, and within each spectrum, the relative heights of the lines are to be proportional to the number of protons responsible for each of the predicted absorptions. Do not skew multiplets. The predicted spectrum of ethanol is shown below as an example of what is expected.
4. The infrared and 1H nmr spectra of a compound with molecular formula C6H11O2Cl are shown below. Deduce the structure of the compound from these data.
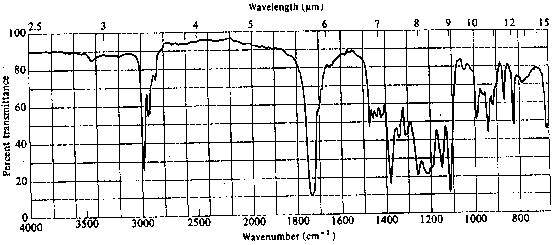
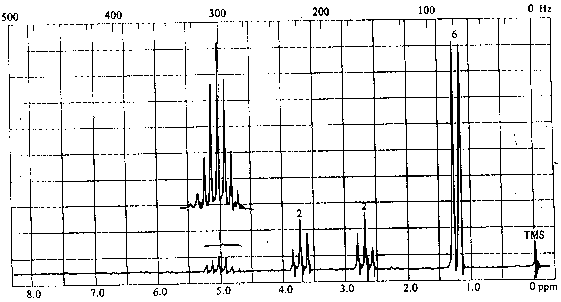
5. The infrared and 1H nmr spectra of a compound with molecular formula C4H7O2Cl are shown below. Note that the multiplet at d~2 ppm in the nmr spectrum is a pentet. Deduce the structure of the compound from these data.
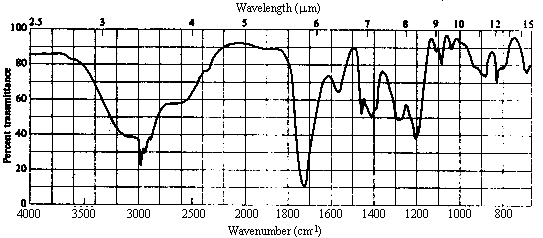
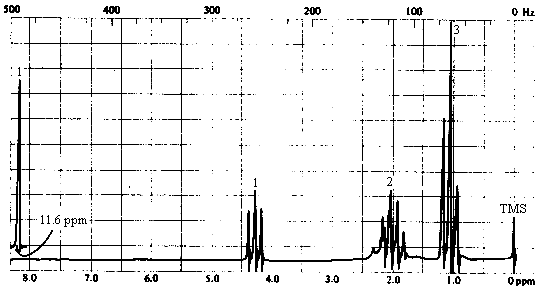
6. The following incomplete reaction sequences give only a starting material and a product that can be prepared from it by a multistep reaction sequence. Show the reagents that would that would be necessary to carry out each transformation, and any major products that would be formed in the intermediate stages.
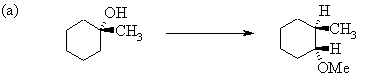
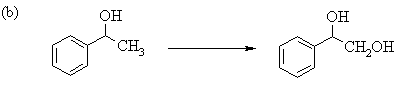

Make sure that your answers are STAPLED TOGETHER and labelled with:
Your NAME & STUDENT NO.
Your TUTORIAL LEADER
Your LAB-SECTION/GROUP/TA (Repeaters should indicate their Tutorial Section)
Assignments are due at 5pm on Friday, January 9 (in your Tutorial Leader's DropBox in ABB). Late assignments will receive a grade of ZERO, unless a medical slip is filed with the Dean's Office.
| Go to: | Instructions for Printing this Document Chem2O6 Problem Sets & Answers Chem2O6 Home Page. |
07dec97; wjl