Chem2O6
- 1997/98| Problem Set #3 | September 26, 1997 |
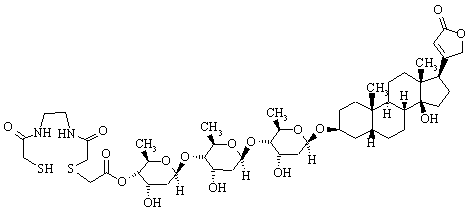
1. The digitoxin derivative shown below is a metal chelating agent that is specific for ATPase and has potential use in radio-imaging.

(i) Identify the functional groups present in this compound.
(ii) Identify a methyl group, and a primary, secondary, or tertiary carbon
(if there are any present).
(iii) Identify a primary, secondary, or tertiary alcohol (if any are present).
(iv) Identify a primary, secondary, or tertiary amide (if any are present).
2. Consider the following three organic molecules:
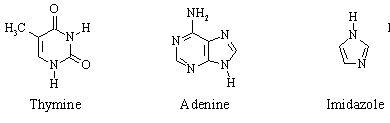
(a) Give the hybridization of each of the atoms (excluding hydrogens) in each molecule.
(b) In which direction do the lone pairs on the N atoms in the three structures point relative to the plane of the rings?
(c) A cyclic array of 6 ![]() electrons (or 6
electrons in overlapping 2p orbitals is said to be aromatic:
electrons (or 6
electrons in overlapping 2p orbitals is said to be aromatic:
(i) Would you expect thymine, adenine, or imidazole to be
aromatic? If so, what resonance structures would you have to write in order to obtain 6 ![]() electrons in a cycle?
electrons in a cycle?
(ii) The first sites of protonation of adenine by a strong acid
(e.g. HCl) are N(1), N(3), and N(7), NOT the NH2 group as one
might expect. Write one or more resonance structures that would account for this.
3. Show the direction (LHS or RHS) that you would expect the following equilibria to lie:
(i) CH3C N + C6H5-
N + C6H5- 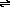 [CH2C
[CH2C N]- + C6H6
N]- + C6H6
(ii) CH2CH2 + OH- 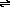 CH2CH-
+ H2O
CH2CH-
+ H2O
(iii) OH- + CH3NH2 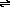 CH3NH-
+ H2O
CH3NH-
+ H2O
(iv) CH3SH + CH3- 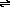 CH3S-
+ CH4
CH3S-
+ CH4
(v) CH3CH2O- + C6H5COOH 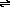 CH3CH2OH + C6H5COO-
CH3CH2OH + C6H5COO-
(vi) (C2H5)2NH2+ + HCO2-
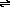 (C2H5)2NH + HCO2H
(C2H5)2NH + HCO2H
4. The following equilibria all lie on the right hand side of the equation:
NH2- + CH3COOCH3 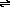 NH3 + -CH2COOCH3
NH3 + -CH2COOCH3
-CH2COOCH3 + CH3COCH3
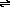 CH3COOCH3 + -CH2COCH3
CH3COOCH3 + -CH2COCH3
-CH2COCH3 + CH3OH 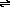 CH3COCH3 + CH3O-
CH3COCH3 + CH3O-
CH3O- + CH3NO2 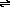 CH3OH + -CH2NO2
CH3OH + -CH2NO2
(i) Identify which compounds are weaker acids than methanol.
(ii) Identify which compounds are weaker bases than methoxide ion.
(iii) Identify which compounds are weaker acids than acetone (2-propanone).
(iv) Identify which compounds are weaker bases than acetone anion.
(v) Identify which is the strongest acid and which is the strongest base.
| Go to: | Instructions for Printing this Document Answers Chem2O6 Problem Sets & Answers Chem2O6 Home Page. |
25sep97; wjl